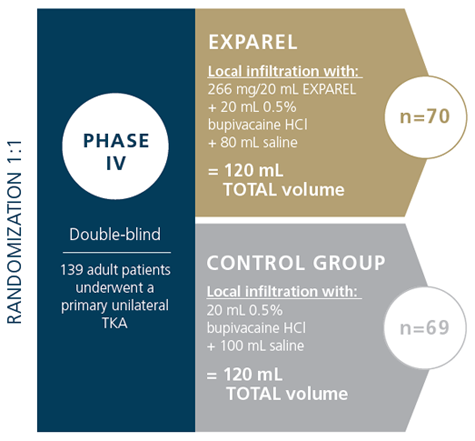
The PILLAR study assessed the safety and effectiveness of EXPAREL vs bupivacaine HCI in total knee arthroplasty (TKA) using standardized volume, technique, and protocol, which included opioid minimizing multimodal pain management in both the study and control groups.
STUDY DESIGN1
- 16 study sites in the United States enrolled 139 patients
- All patients were required to stay in the hospital for 48 hours following surgery to capture all data required to assess primary endpoints
- All surgeons were trained on and were required to follow a standard infiltration technique and protocol, as described in the published administration protocol for TKAs
Primary endpoints:
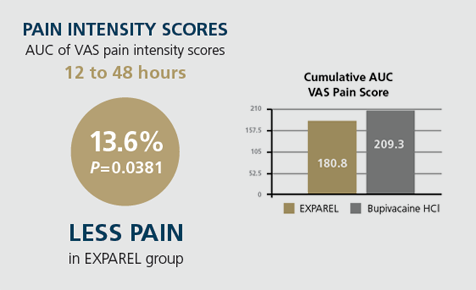
- Area under the curve (AUC) of visual analog scale (VAS) pain intensity scores 12 to 48 hours post surgery
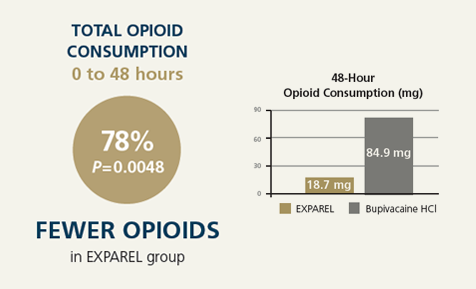
- Total opioid consumption 0 to 48 hours post surgery
Secondary endpoints:
- AUC VAS pain scores through 72 hours
- Opioid consumption through 72 hours
- Percentage of patients opioid-free through 72 hours
- Time to first opioid rescue through 72 hours
Safety endpoints:
- Treatment-emergent adverse events (TEAEs)
- Serious adverse events
- Opioid-related adverse events
- TEAEs of special interest (cardiac events, neurologic events, and falls)
STUDY RESULTS
Using the PILLAR protocol and administration technique, surgeons achieved consistently favorable outcomes with EXPAREL compared with bupivacaine HCl
- Both treatments were well tolerated, and EXPAREL demonstrated a safety profile similar to that of bupivacaine HCl

STUDY PROTOCOL AND TECHNIQUE
To achieve optimal analgesia, it is suggested to administer EXPAREL using:
- Consistent volume (120 mL) mixed with free bupivacaine HCl
- Consistent infiltration technique at anatomical sites with high nerve density (1 to 1.5 mL volume spaced 1 to 1.5 cm apart)
- Opioid-minimizing multimodal pain management protocol following surgery
References
- Mont MA, Beaver WB, Dysart SH, Barrington JW, Del Gaizo DJ. Local infiltration analgesia with liposomal bupivacaine improves pain scores and reduces opioid use after total knee arthroplasty: results of a randomized controlled trial. J Arthroplasty. 2018;33(1):90-96. doi:10.1016/j.arth.2017.07.024.

